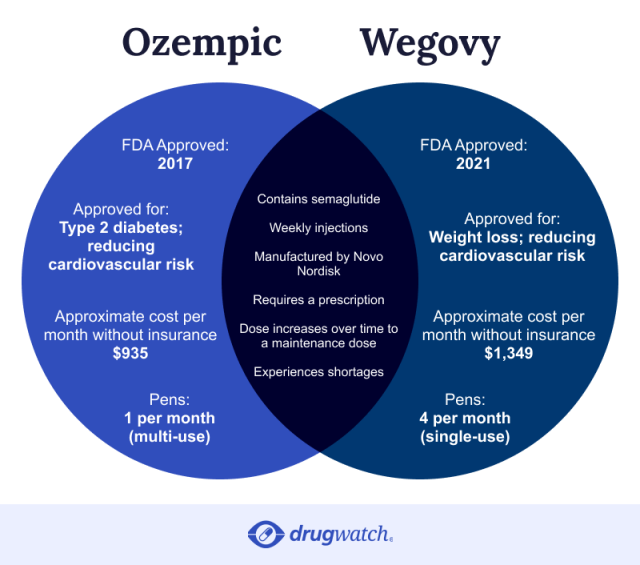
Higher Dose of Wegovy Approved for Enhanced Weight Loss
The U.S. Food and Drug Administration (FDA) has recently approved a higher dose of the weight loss medication Wegovy, which promises to help individuals achieve greater weight loss results. This new formulation, known as Wegovy HD, delivers a dosage of 7.2 milligrams per week via injection, significantly higher than the previously available 2.4 milligrams.
Novo Nordisk, the company behind Wegovy, developed this stronger dose in response to findings that some patients were unable to meet their weight loss goals with the lower dosage. In a clinical trial conducted in 2025, known as the STEP UP phase 3b trial, participants given the higher dosage experienced an average weight loss of approximately 18.7%. Notably, around one-third of these participants shed at least 25% of their body weight.
The FDA’s swift approval of this higher-dose version demonstrates a commitment to advancing public health initiatives. The agency granted approval just 54 days after the application was submitted, marking it as the fourth product to receive approval under the Commissioner’s National Priority Voucher pilot program.
FDA Commissioner Martin Makary expressed optimism about the approval process, highlighting the efficiency with which the FDA is now operating to support innovations that align with national health priorities.
While the potential for increased weight loss is promising, the higher dosage may also come with risks. Clinical data suggests that the safety profile of the 7.2 mg dose aligns with known side effects of semaglutide, the active ingredient in Wegovy. Common side effects include gastrointestinal issues such as nausea, diarrhea, constipation, vomiting, and abdominal pain. Patients may also experience altered skin sensations at higher doses, although these effects typically diminish over time or with dose adjustments.
There is cautious optimism among health professionals regarding the new dosage. Dr. Meghan Garcia-Webb, an expert in obesity medicine, noted the importance of monitoring patients closely, especially given recent studies indicating a possible increased risk of ischemic optic neuropathy (ION) with higher doses. This condition, often referred to as an “eye stroke,” can lead to sudden vision loss or blindness and may be more prevalent in male patients. Dr. Garcia-Webb emphasized the need for patients to consult their ophthalmologists if they have pre-existing eye conditions before starting treatment with Wegovy.
As patients consider Wegovy as a weight loss option, they should be aware of the potential side effects, which also include headaches, fatigue, and low blood sugar in those with type 2 diabetes. While many side effects are generally mild, it’s essential for individuals to discuss any concerns with their healthcare providers.
In addition to side effects, cost and the necessity for long-term use to maintain weight loss should also be taken into account when evaluating Wegovy as a treatment option. As more data emerges on the higher dose, both patients and healthcare professionals will be better equipped to make informed decisions regarding its use in weight management.