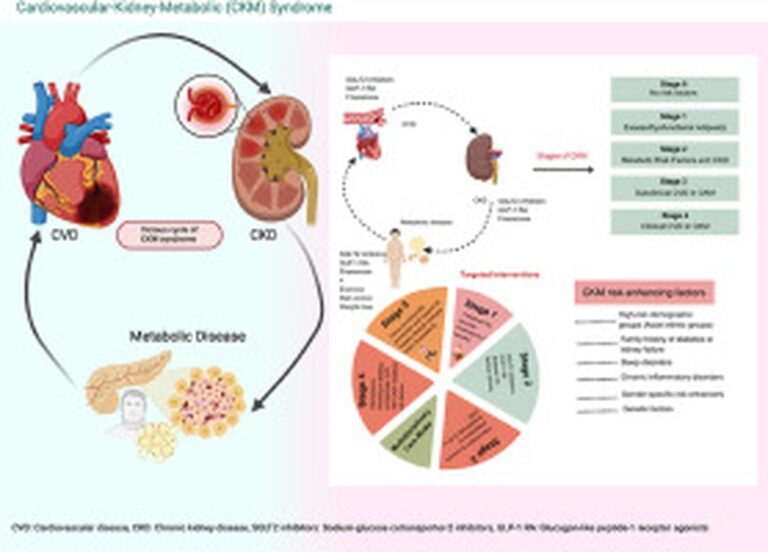
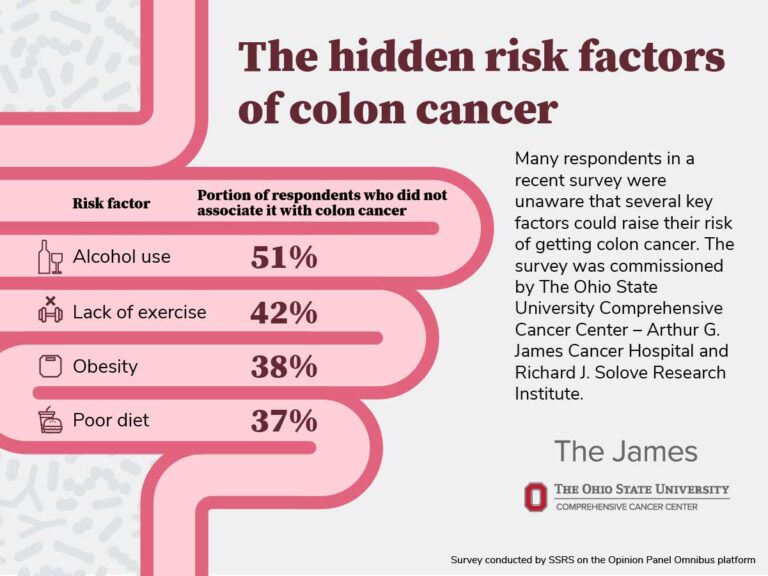
New GLP-1 Drug Survodutide Shows Promising Weight Loss Results
A new weight loss medication, survodutide, has emerged as a potential game-changer in the fight against obesity, according to findings from a recent Phase 3 clinical trial conducted by Boehringer Ingelheim. The trial demonstrated that this innovative drug could lead to an average weight loss of 16.6% among participants, significantly outperforming the placebo group, which recorded only a 3.2% reduction.
Survodutide works by stimulating both glucagon-like peptide-1 (GLP-1) and glucagon receptors, a dual-action approach that may enhance metabolic health and support weight loss efforts. This clinical trial, known as SYNCHRONIZE-1, involved 725 adults with obesity or overweight who did not have type 2 diabetes, and it spanned 76 weeks across multiple international sites.
The results of the trial were notable not only for the amount of weight lost but also for improvements in waist circumference, a key indicator of metabolic health. Participants reported a significant decrease in waist size, which is associated with reduced cardiometabolic risk. The findings suggest that survodutide could be beneficial not just for weight loss but also for addressing obesity-related conditions like liver disease.
While survodutide is similar to other GLP-1 receptor agonists, such as tirzepatide (known by the brand name Mounjaro), it uniquely activates glucagon receptors, which may aid in regulating liver metabolic function. This is particularly important as it has the potential to decrease liver fat and inflammation, which are critical factors in many obesity-related complications.
In the trial, participants received weekly injections of either 3.6 or 6.0 milligrams of survodutide or a placebo. Remarkably, 85.1% of those treated with the drug achieved at least a 5% reduction in body weight, compared to just 38.8% in the placebo group. On average, participants lost about 39.2 pounds, primarily from fat rather than lean muscle.
The side effects reported were predominantly mild to moderate, including gastrointestinal issues like nausea. These occurred mainly when participants were adjusting to the drug dosage. Importantly, no new safety concerns were identified throughout the duration of the trial.
Experts are cautious about labeling survodutide as a definitive breakthrough in weight loss medications. Dr. Hector Perez, a lead bariatric surgeon, emphasized the need for comprehensive data before drawing conclusions about its efficacy compared to existing treatments. However, he acknowledged the potential of survodutide to address not only weight issues but also underlying conditions like fatty liver disease due to its dual-action mechanism.
As the medical community awaits further details, including a full presentation of the trial data at the upcoming American Diabetes Association conference, individuals interested in weight loss treatments are encouraged to discuss their options with healthcare professionals. This includes comparing potential side effects, considering existing GLP-1 medications, and evaluating eligibility for other interventions, such as bariatric surgery.
In summary, while survodutide shows significant promise for weight loss and metabolic health, further evaluation is necessary before it can be fully endorsed as a treatment option.